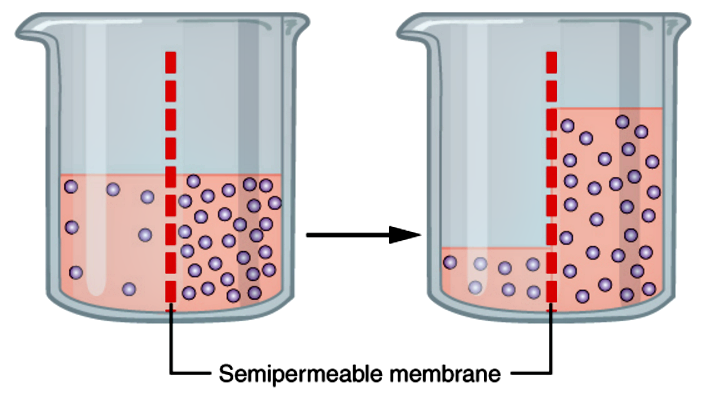
Osmosis
Osmosis is the diffusion of water molecules across a semipermeable membrane from an area of higher concentration to and area of lower concentration. A solution can be described in three ways. Hypertonic, isotonic, and hypotonic. Hypertonic is when a solution has a higher concentration than a cell. So water flows outside the cell, causing it to shrivel and even die. Isotonic is when a solution to a cell has the same concentration of dissolved particles as the cell. Water molecules move in and out of the cell at an even rate, keeping the size constant. Hypotonic is when a solution has a lower concentration than a cell. Water molecules are more concentrated outside the cell, so water diffuses into the cell and the cell membrane expands, until it could probably burst. It all has to do with what’s more concentrated, the solution, or the cell.
Osmosis has a lot more to do with how plant and animal cells work. Some animal cells and single-celled organisms can survive in a hypotonic environments. Their cells have adaptations for removing excess water. In plants the rigid cell wall prevents the membrane from expanding too much. It is important to recognize that the higher concentration of particles in a dissolved solution, the lower the concentration of water molecules in the same solution. The decision about which side of the membrane to call “high” osmotic pressure is a difficult one. Osmotic pressure is the pressure that would have to be applied to a pure solvent to prevent it from passing into a given solution by osmosis. The choice that has to be made is a little bit the opposite of what textbooks say, which attribute “high” osmotic pressure to the solution and no osmotic pressure to pure water. The rational for the choice is that the energy that drives the fluid transfer is the thermal energy of the water molecules. Plus that density is higher in the pure solvent since there are more water molecules. The thermal energy of the solute doesn’t contribute to the transport, which presumes the membrane is impermeable to them. Moving on, pure water has the actually has the highest concentration possible. As more salt or sugar is dissolved the water concentration decreases. A concentration gradient is where there is a high water concentration in one area and a lower one in another.